Operating Diagram Characteristics

1.- The electrodes separate the hydrogen from the oxygen in the lower tank by electrolysis. This is done by electrodes, which are platinum coated titanium grills, one connected to a positive pole in contact with the water in the lower tank, and the other connected to a negative pole in contact with the upper water, in the base of the jug.
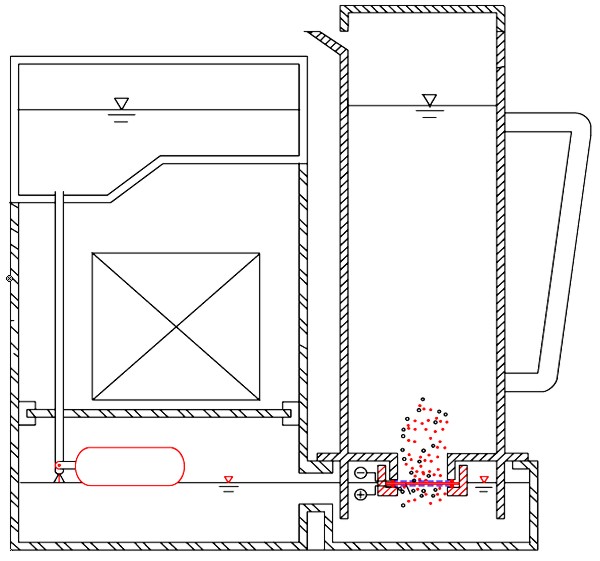
2.- There is a membrane between both electrodes, which only allows hydrogen ions through, as they are much smaller than oxygen ions.

Hydrogen ions are hydrogen atoms without electrons, which gives them a positive electrical charge. These are attracted by the negative pole above the membrane, which causes them to cross through into the jug.
3.- Once the hydrogen ions are in the jug, they take the electrons from the negative pole, converting them into “active hydrogen” atoms.
CREATION OF HYDROGEN BUBBLES IN THE BASE

We’ll call you back
Tell us your telephone number and a time and we’ll call you back and offer you the best personal service.